Reading Navigation
How Many Ketamine Treatments for Depression Are Needed?

The question on all the ketamine researchers’ lips right now is around the idea that if ketamine can lift depression in hours, why isn’t one treatment enough? How do we extend the benefits? Our culture and attention spans are wired for quick fixes… from microwaved mac-n-cheese to the three-second TikTok video. It’s nice to imagine that a single infusion of ketamine could act as some sort of “neurological reset.” Unfortunately, ketamine doesn’t “cure” depression in one fell swoop. Ketamine opens a brief window that often closes without additional support. The question this article seeks to answer is not whether ketamine works, per se. But rather, how many times does the brain need that ketamine window to open for change to stick around?
Ketamine’s complicated origin story
You might have heard of ketamine, but maybe not in this context.
Depending on your age, social circle, or social-media algorithm, you may have heard of ketamine in the context of a “party drug” (Special K, K, Kit Kat, Vitamin K, cat tranquilliser… did we just date you?). You might also recognise it from the operating room, where ketamine has been used in surgeries as a dissociative anaesthetic. Maybe you have filmed a loved one waking up post-op, speaking nonsense or describing feeling outside their body? Different settings, same effect: ketamine unfailingly kicks your brain out of autopilot mode and into a dreamlike state.
Ketamine was first synthesised in 1962 as a fast-acting general anaesthetic. It was actually used frequently as a battlefield medicine1 during the Vietnam War. Medics used it to keep soldiers alive until they reached more comprehensive care. Ketamine’s dissociative effects inadvertently made soldiers feel detached from pain, less aware of injuries, and less overwhelmed by panic. Outside of the war, though, ketamine was developing a “sus” reputation. Party-drug lore fueled stigma about hallucinations and out-of-body experiences. During this period, ketamine’s psychiatric effects were ignored, and the field of psychiatry was not buying it.
How depression science outgrew the “monoamine model”
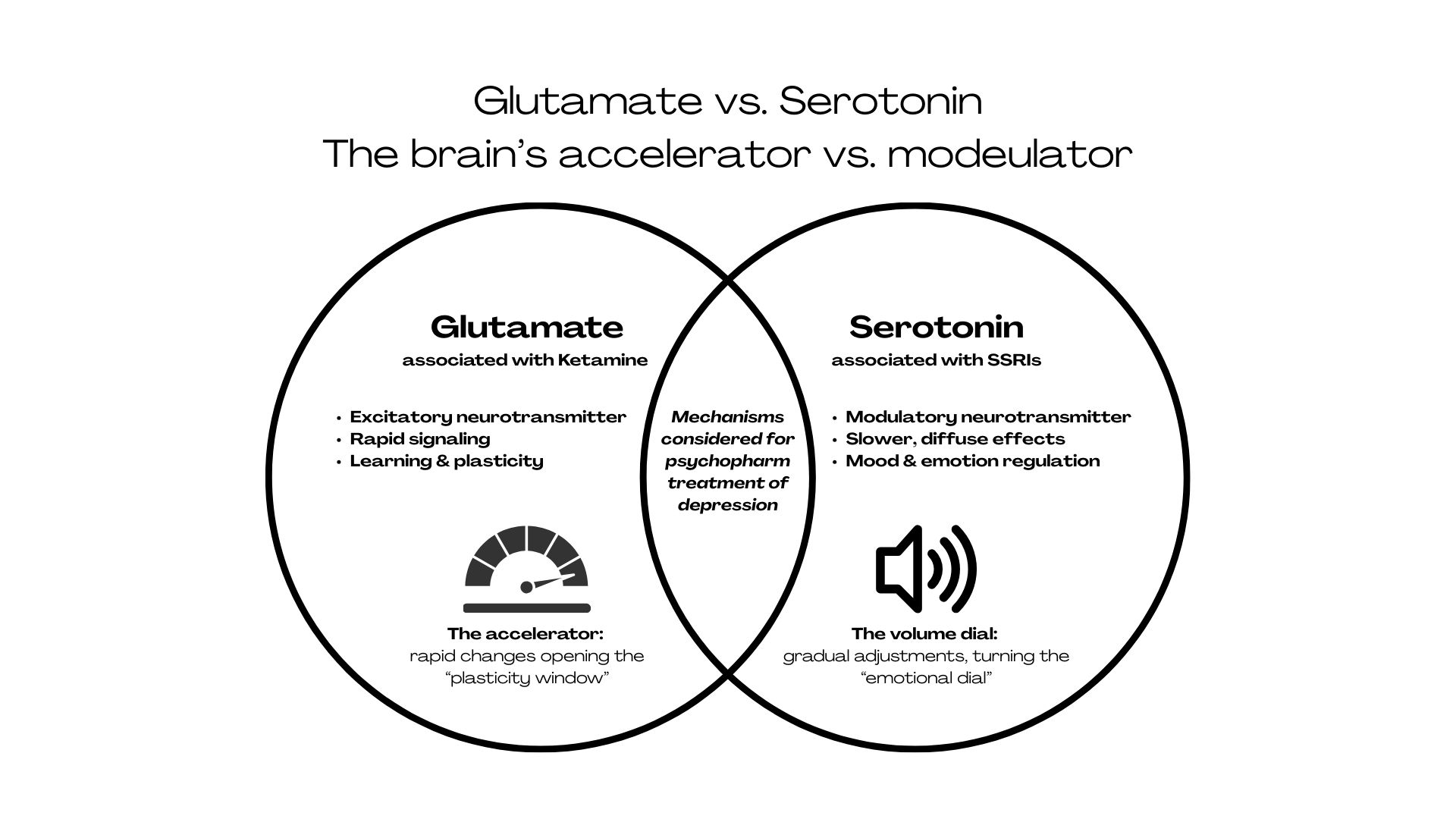
In a parallel process, during the 1990s, neuroscience slowly questioned the long-held idea that depression was specifically a problem with serotonin. Serotonin was painted as the key neurotransmitter involved in mood regulation and considered the main target for traditional antidepressants (i.e., Selective Serotonin Reuptake Inhibitors (SSRIs)). These medications fall under the monoamine model of depression, which presumes that depression stems from deficits in neurotransmitters like serotonin, norepinephrine, and dopamine.
SSRIs have helped many, but patients and providers alike complain that they can take weeks or months to work and cause unwanted side effects. Depression researchers were unsatisfied2 with a medication that was often considered not much better than a placebo. Attention was opened to other hypotheses, including one involving glutamate and brain plasticity3 (the brain’s capacity to change and adapt). Glutamate is the brain’s primary excitatory neurotransmitter, influencing learning, connection, and neural flexibility. And here is where we bury the lede; this is where ketamine comes in.
The 2000 turning point
In 2000, the first randomised, placebo-controlled trial4 (a gold-standard study design that randomly assigns participants to treatment or placebo and compares outcomes) showed low-dose intravenous (IV) Ketamine offered fast-acting antidepressant effects… improvements within hours. This finding cracked the door open to a new way of thinking about treatment for depression.
In the early to mid-2000s, small NIH-funded studies5 replicated the original findings, with promising effect sizes for treatment-resistant depression and suicidal ideation. The field gained momentum in the 2010s, with larger trials and specific dosing protocols6. During this period, clinics began offering off-label IV ketamine7, and media coverage quickly labeled it a “miracle drug,” setting the bar high.
In 2019, the FDA approved esketamine nasal spray8 for treatment-resistant depression, which helped legitimise the field. Intranasal esketamine is not the same as IV ketamine: indications, dosing schedules, and monitoring requirements differ by route and regulator. In many systems, it’s delivered in a supervised clinic setting with a structured dosing schedule, which can influence how ‘number of sessions’ is planned compared with off-label IV protocols. Fast forward to the 2020s, and there has been an explosion of Ketamine clinics, integration models, therapy pairing, trauma-informed approaches, and debates about ethics, access, and long-term outcomes. Each of these subjects could be a rabbit hole, but for now, let us return to the basics of ketamine.

A Turning Point for Your Mental Health
Unsticking the depressed brain
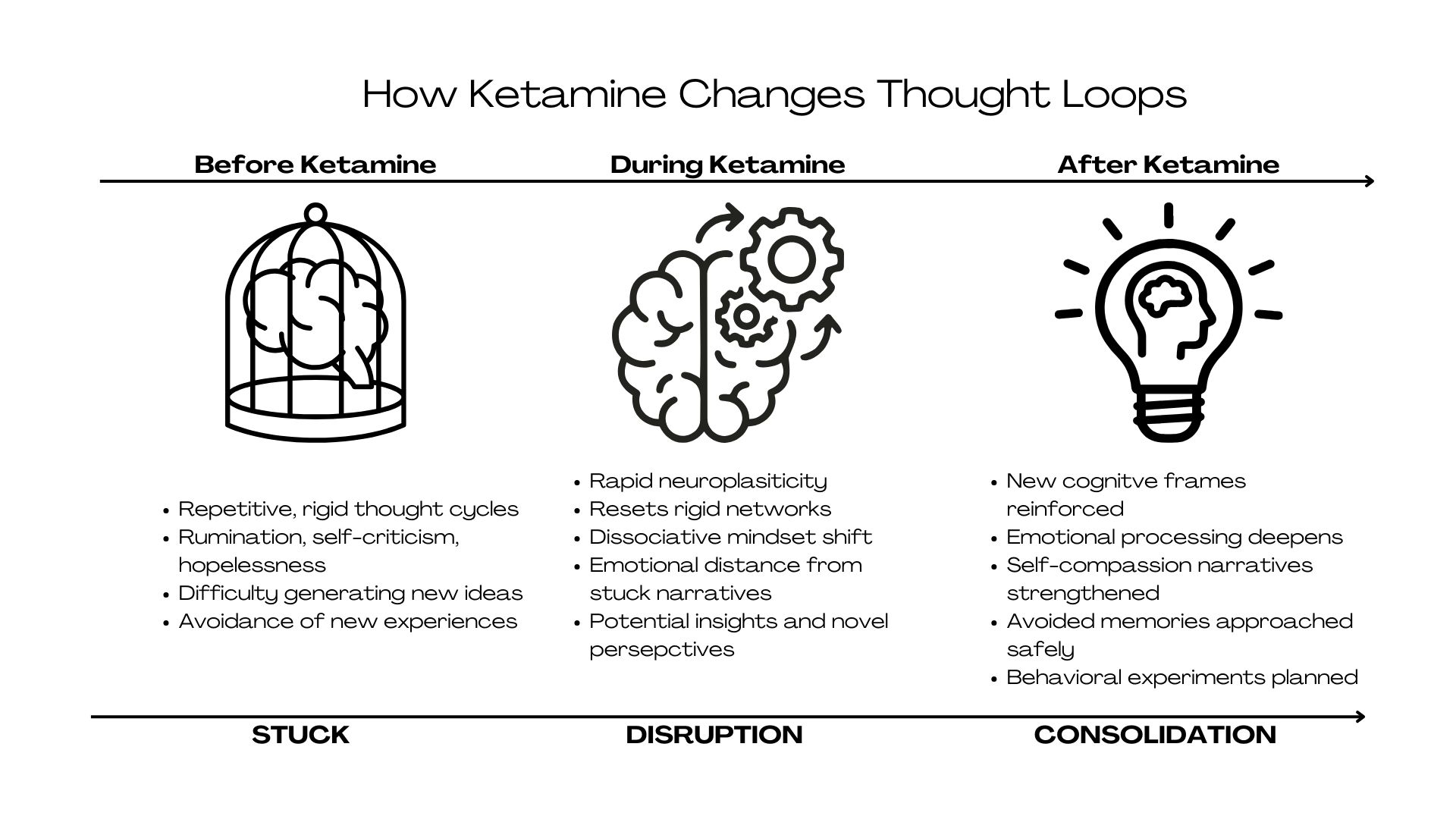
From a bird’s-eye view, ketamine works by “unsticking” the brain or, more scientifically, rapidly increasing neuroplasticity in circuits that become rigid and over-inhibited in depression, particularly those associated with the Default Mode Network (DMN)9. The DMN is efficient, giving the brain the power to rely on automatic patterns without tons of conscious effort. Think, for example, how you don’t have to think so hard at a red light to stop, because your brain’s DMN is running on autopilot; it saves you lots of mental energy!
In depression, though, this system can become overactive, often defaulting to the same unhelpful predictions and self-stories that keep you stuck in patterns you desperately want to break. Ketamine temporarily loosens these automatic responses, creating space for new ways of thinking.
At a neurochemical level, ketamine blocks NMDA receptors involved in glutamate signaling. Downstream effects may include short-term changes in synaptic signaling and connectivity in networks linked to rumination and cognitive control. These are useful models for understanding why some people feel ‘unstuck,’ but the exact pathways driving antidepressant response are still being clarified.

What the experience actually feels like
So, what does Ketamine feel like? People often notice changes in both the body and consciousness (what scientists have dubbed “dissociation”). Some researchers see dissociation as a side effect, while others see it as a key mechanism of change (it is still largely debated at the coffee station of big psychedelic science conferences).
During ketamine treatment, individuals may feel detached from their usual sense of identity and perception and be more able to observe their experience rather than feel overwhelmed by it. In the body, this can feel like a sense of lightness or heaviness, reduced pain or tension, altered perception of time or space, or a feeling of being less “located” in the body. It is quite difficult to describe without experiencing it.
Why repetition matters
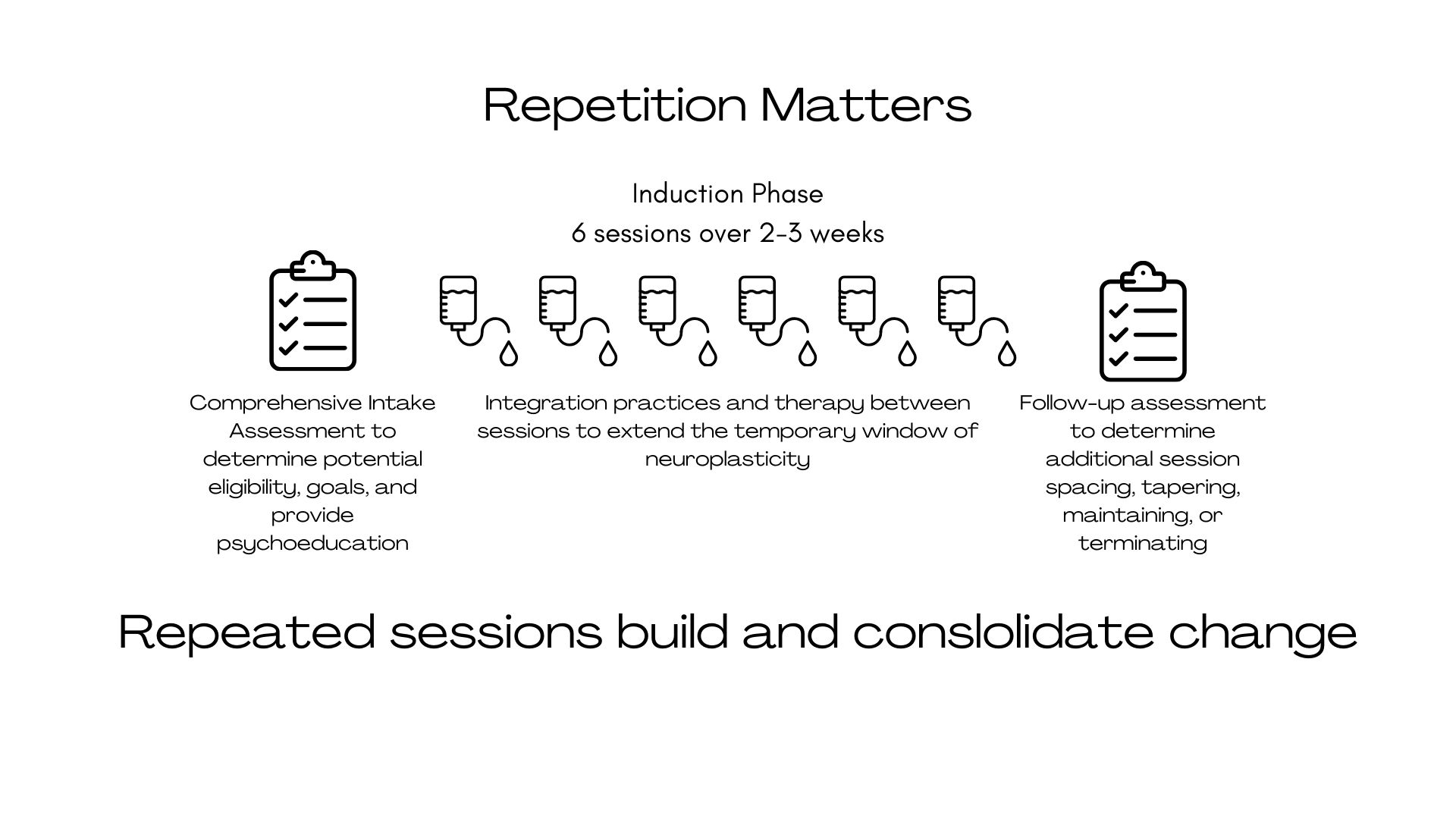
In contrast to traditional antidepressants, which may take weeks to exert effects, ketamine can produce meaningful mood improvements within a day, highlighting its potential utility for acute symptom relief in treatment-resistant depression. In fact, individuals report that a single session can reduce symptoms5 quickly, but those effects often fade within days. Instead, research has demonstrated that benefits accumulate10 and become more durable with repeated treatments11 rather than a single dose.
Ketamine treatment is often offered as a short series of six sessions over two to three weeks, with therapy and integration practices between them. Phillips et al. (2019) showed6 that the six infusions administered over 12 days can lead to a significant cumulative response in treatment-resistant depression, and this protocol has influenced induction templates used in many clinics. Singh et al. (2016) further compared11thrice-weekly vs twice-weekly dosing, with twice-weekly (0.5 mg/kg over 40 min) showing similar maintained efficacy over ~15 days, helping inform common induction schedules used in many clinics.
Finally, the APA Consensus Statement on Ketamine for Mood Disorders12 was created, underscoring the two-to-three-week infusion model, but mentioned uncertainty about the optimal maintenance schedule. After the first phase of six sessions, additional treatments can be spaced, tapered, or moved to as needed based on one’s response.
While scientists broadly agree that ketamine has antidepressant effects and clinicians lead with the six-session induction model, many still debate how best to extend ketamine’s effects. Some emphasise timing and session count (as discussed above), while others prioritise therapy, and many highlight both. Ultimately, Ketamine opens the window, but it doesn’t tell the brain and body what to learn. Therapy can provide13 the structure for exploring new meaning and perspectives and shift insight14 into behaviour change.
To continue Rick’s metaphor, ketamine can create fresh powder, but therapy and integration decide where to put the new tracks.
Who is ketamine for?
Ketamine is considered helpful15 for people with treatment-resistant depression (TRD), major depressive disorder with significant functional impairment, suicidal ideation, or those who have engaged in therapy but continue to feel “stuck”.
It is not often considered a first-line treatment. TRD is typically defined16 as an inadequate response to two or more antidepressant trials at an adequate dose and duration and often involves severe, persistent symptoms, longer illness duration, higher rates of comorbidity with other medical and mental health conditions, and significant trauma exposure or functional impairment.
Importantly, TRD is not a personal failure; it reflects the complex interaction of biology, stress, trauma, and social context. Ketamine has also shown promise in rapidly reducing suicidal ideation17 when other treatments move more slowly.
Ups and downs are part of the process
Responses to ketamine vary. This variability is expected18. Some people notice improvements in mood or suicidal thoughts within one or two sessions, but these gains often fade without additional treatment. Others improve more gradually, with shifts in energy, perspective, and emotional range coming before any lasting mood changes; these individuals may benefit from psychotherapy and integration practices. Partial responses are also common, such as reduced suicidality without mood improvement. This also may indicate the need to strengthen therapeutic support or address co-occurring mental health disorders rather than cease treatment.
During the initial six sessions, symptom fluctuations are normal. Some days may feel shockingly better, while others may feel flat, foggy, or emotionally tender. As clinicians, we often see this as a reflection of increased emotional access (minimising avoidance) rather than clinical decline. Progress in any mental health treatment is rarely linear and often looks and feels clumsy. Completing the full series of six sessions is usually important, as early improvements often fade if treatment is stopped too soon (think: you would not stop an antibiotic early).
Explore Progressive Treatments with Trusted Therapists
A booster or a break?
For some individuals, maintenance or booster treatments can improve gains19 by reopening those windows of plasticity and reinforcing learning; however, there is no one-size-fits-all protocol. Longitudinal follow-up data20 from repeated subanesthetic infusion protocols suggest that while a meaningful proportion of patients maintain antidepressant response and functional improvement for months after an acute series, relapse most commonly occurs within the first few weeks following cessation, highlighting a clinically sensitive period where monitoring and support are important.
After the initial course, providers should continue monitoring symptoms and functioning, and they may recommend pausing, spacing, maintaining, or discontinuing treatment. Decisions about boosters are informed by patterns of symptom return, patient goals, stress load, response to psychotherapy, and lifestyle or behavioural factors. As you can probably pick up on by this point, the most effective care combines ketamine with psychotherapy and integration to help insights translate into lasting change.
Various routes of administration
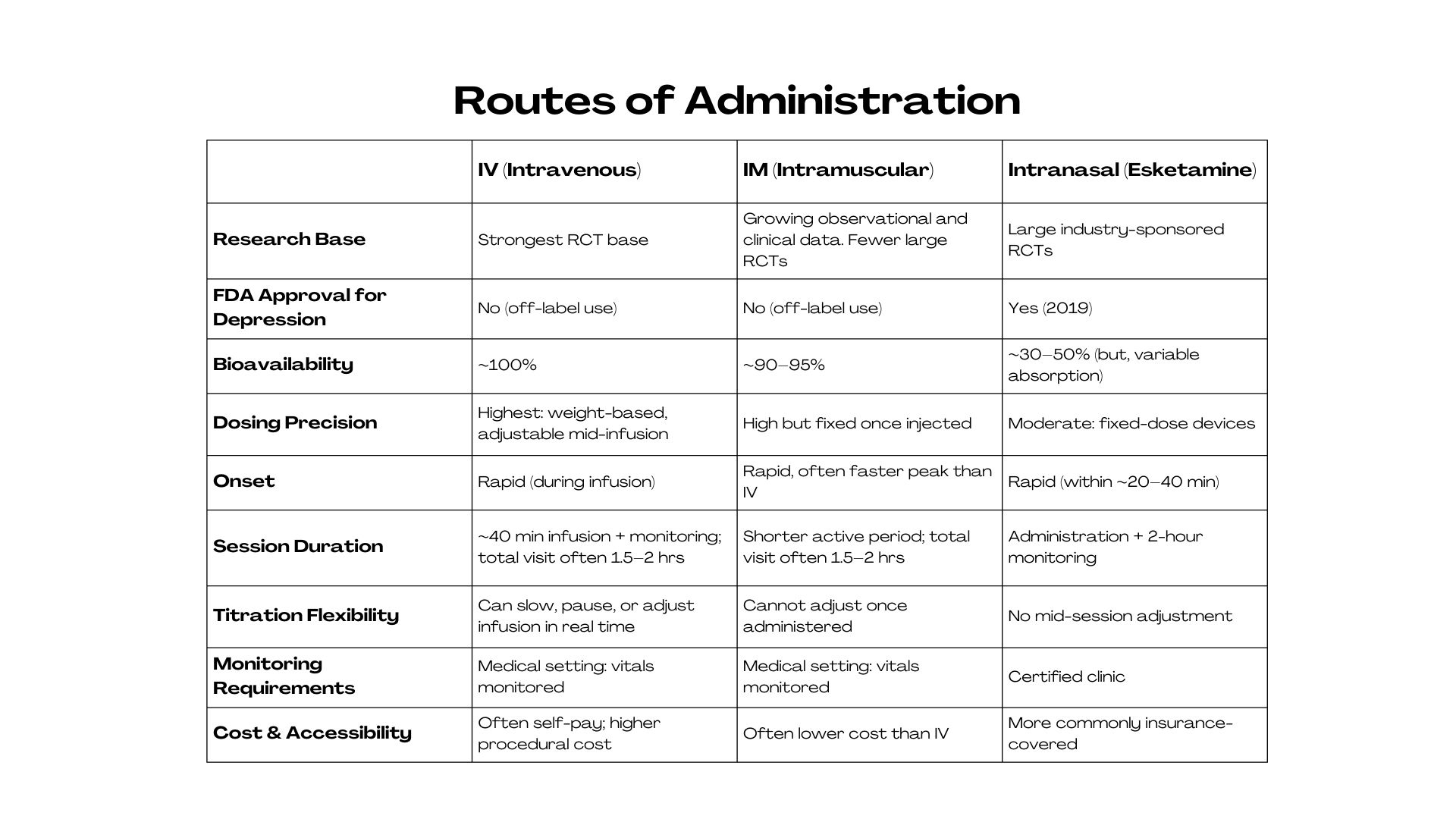
There are several routes of ketamine administration21, each with pros and cons. Because ‘ketamine therapy’ can mean different routes (IV, IM, intranasal esketamine, or compounded oral/sublingual forms), the evidence base and oversight differ. Session number and spacing depend heavily on route, setting, and how closely treatment is monitored.
- Intravenous (IV) ketamine is considered the gold standard in research, as it allows for precise dosing delivered through a slow, controlled infusion (over about 40 minutes). It has a rapid onset and a high degree of control, but it requires a clinical setting with medical monitoring.
- Intramuscular (IM) ketamine is also commonly used. Ketamine is injected into a muscle, leading to a faster onset and shorter session. While dosing is slightly less precise than IV administration, it may be a good fit for individuals who tolerate stronger peak experiences or for settings without infusion capabilities.
- Intranasal ketamine, including FDA-approved esketamine, is administered under medical supervision22. It is less invasive and more accessible, particularly in insurance-based care models, and is sometimes used for ongoing or maintenance treatment23. Absorption can be more variable, but it offers a more practical option for many.
Side effects
Temporary side effects during ketamine therapy are typically short-lived6. These may include dissociation during or after sessions, nausea, headache, fatigue or grogginess, and brief increases in anxiety or blood pressure.
Screening and contraindications
Ketamine therapy is not appropriate for everyone, which is why thorough screening matters. It may be contraindicated12 for individuals with uncontrolled high blood pressure or cardiovascular disease, certain neurological conditions, active psychosis or unmanaged bipolar mania, severe and uncontrolled substance use disorders, or during pregnancy, depending on the provider. Individuals with severe or complex posttraumatic stress disorder may also be more vulnerable24 to emotional destabilisation if treatment is not thoughtfully scaffolded.
Addiction risks
There is also an important conversation about addiction risk. Ketamine can produce reinforcing effects that may lead to tolerance and misuse, and the risk varies by individual history. Supervised programs reduce risk through careful screening, monitoring, and spacing/limiting treatment — especially if benefits plateau or a person starts to feel they ‘need’ ketamine. Ketamine is sometimes considered more psychologically addictive than chemically addictive. Concerns arise if someone begins to feel they “need” ketamine or view it as the “only path” to healing. Importantly, the clinical use of ketamine occurs within a structured medical framework that differs substantially from recreational contexts, where dosing variability, polysubstance use, and lack of psychological support introduce risks.
Safety!
When delivered in medically supervised, evidence-based settings, ketamine is considered safe and well-tolerated for carefully screened patients. Vital signs are often monitored during treatment, and dosing is controlled. In clinical settings, long-term risks appear low in monitored clinical protocols, though optimal maintenance frequency and long-term outcomes remain areas of active study.
The various costs
Ketamine treatment can require a big investment of time, money, and emotional energy, which can be challenging alongside other responsibilities. Costs vary depending on the route of administration (IV, IM, or intranasal), geographic region, clinic model, and level of medical and therapeutic support. IV or IM treatments can cost several hundred dollars per session, while intranasal esketamine may be partially covered by insurance. Providers must clearly explain pricing, outline the expected number of sessions, and discuss alternatives. You can explore the UK’s offerings here.
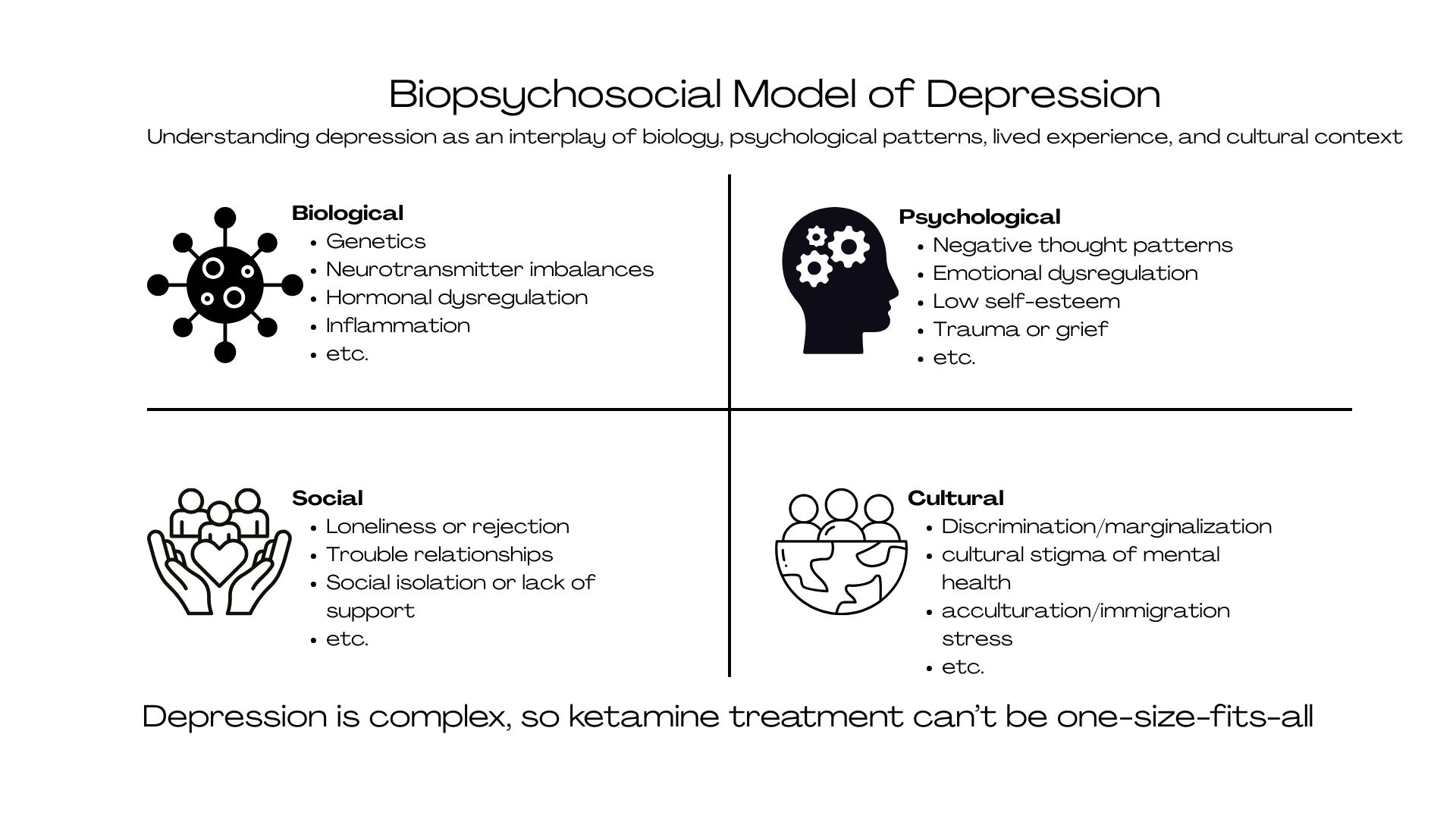
Depression is multifaceted
To close, depression is best understood through a biopsychosocial lens, or an interaction between brain biology, psychological patterns, and lived context. Diagnosis in the mental health world tells us how the disorder is expressing itself, not why it developed (defined by clusters of symptoms, not underlying causes). In other words, a diagnosis of depression is not an X-ray.
This distinction matters for ketamine treatment since ketamine does not target a diagnostic label, but rather neurobiological rigidity (a process that is common in many forms of depression and other mental health struggles). Co-occurring conditions (e.g., anxiety disorder, trauma-related symptoms, substance use, personality patterns) can influence treatment response25, as overlapping neurobiological and psychological processes can shape intensity and durability of ketamine’s effects.
Ketamine’s effectiveness depends not only on the drug itself, but on how these biological shifts are integrated psychologically, a process impacted by comorbidities. When ketamine is embedded within comprehensive assessment, psychotherapy, and multidisciplinary care, it aligns with the understanding that depression is dynamic, layered, and, most importantly, treatable.
So… how many treatments?
Because depression reflects a biopsychosocial process rather than a single entity, the number of ketamine treatments is best understood not as a fixed protocol, but as a tailor-made course aimed at sustaining long-term change. For anyone considering ketamine, the goal should always be evidence-based, medically supervised care that prioritizes informed decision-making.
Be cautious of clinics that guarantee results or promise a “cure,” discourage therapy or integration, fail to track symptoms or outcomes, or push overly frequent treatments without regular reassessment. In a medical and mental health landscape keen on fast solutions, long-term outcomes should matter more than quick relief.
Ketamine can work more quickly than other antidepressants, but it doesn’t replace the work of change; it creates the conditions for it.







































